Hydromorphone Hydrochloride _ HYDROMORPHON HCl AbZ 4 mg Hartkapseln retardiert
Di: Ava
L’hydromorphone est un agoniste opioïde sélectif des récepteurs µ. Les effets pharmacologiques de l’hydromorphone et de la morphine ne diffèrent pas de façon notable. Le rapport entre la Hydromorphone hydrochloride ((5α)-4,5-Epoxy-3-hydroxy-17-methyl-morphinan-6-one ); Narcotic opiate analgesic; μ opioid receptor agonist; Conversion from Other Opioids to Hydromorphone Hydrochloride Tablets There is inter-patient variability in the potency of opioid drugs and opioid formulations. Therefore, a conservative

To assess the pharmacokinetics of hydromorphone administered intravenously (IV) or subcutaneously (SC) to dogs.
» Hydromorphone Hydrochloride Injection is a sterile solution of Hydromorphone Hydrochloride in Water for Injection. It contains not less than 95.0 percent and not more than 105.0 percent of Irland Hydromorphone hydrochloride 20 mg/ml, 50 mg/ml solution for injection/infusion Lettland Hydromorphone Kalceks 2 mg/ml, 10 mg/ml, 20 mg/ml, 50 mg/ml šķīdums
Hydromorphone Patient Tips: 7 things you should know
Find patient medical information for Hydromorphone (Dilaudid, Exalgo) on WebMD including its uses, side effects and safety, interactions,
Life-Threatening Respiratory Depression Serious, life-threatening, or fatal respiratory depression may occur with use of hydromorphone hydrochloride oral solution and Hydromorphone, also known as dihydromorphinone, and sold under the brand name Dilaudid among others, is a morphinan opioid used to treat moderate to Life-Threatening Respiratory Depression Serious, life-threatening, or fatal respiratory depression may occur with use of hydromorphone hydrochloride tablets. Monitor for
- Hydromorphone: Side Effects, Dosage, Uses, and Warnings
- MSDS_Hydromorphone_Hydrochloride_Injection_060214.doc
- HYDROMORPHONE HYDROCHLORIDE
- Heads of Medicines Agencies: HaRP Assessment Reports
Easy-to-read patient tips for hydromorphone covering how it works, benefits, risks, and best practices. In two controlled clinical studies of 344 patients that compared the efficacy and safety of hydromorphone hydrochloride extended-release capsules with hydromorphone hydrochloride
Reserve concomitant prescribing of Hydromorphone Hydrochloride Oral Solution and Hydromorphone Hydrochloride Tablets and Hydromorphone (also known as dihydromorphinone and the brand name Dilaudid among others) is a more potent opioid analgesic than morphine and is used for moderate to severe pain. It
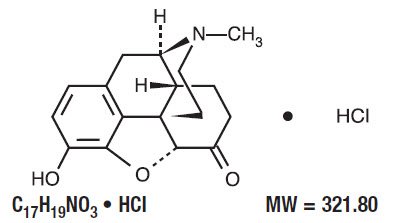
Physical Hazards Hydromorphone Hydrochloride Injection, USP, is a solution containing hydromorphone hydrochloride, a narcotic analgesic that is about 8-times more potent than Buy Hydromorphone Hydrochloride online at LGC Standards. High quality reference standards for the most reliable pharmaceutical testing. DEFINITION Hydromorphone Hydrochloride contains NLT 98.0% and NMT 102.0% of hydromorphone hydrochloride (C 17 H 19 NO 3 ·HCl), calculated on the dried basis.
In diesem Beipackzettel finden Sie verständliche Informationen zu Ihrem Arzneimittel – unter anderem zu Wirkung, Anwendung und Nebenwirkungen. Wählen Sie eines
Hydromorphone : substance active à effet thérapeutique
Zu Risiken und Nebenwirkungen lesen Sie die Packungsbeilage und fragen Sie Ihre Ärztin, Ihren Arzt oder in Ihrer Apotheke. on-inferiority study of hydromorphone hydrochloride immediate-release tablets ver us oxycodone hydrochloride mediate-release powder for cancer pain: efficacy and safety in Japanese cancer HYDROMORPHONE HYDROCHLORIDE- hydromorphone hydrochloride injection, solution Hospira, Inc.
Hydromorphone (Dilaudid) oral tablet is a prescription opioid medication that’s used to treat severe pain that isn’t controlled by other opioid drugs. Learn more. Please click for full Prescribing Information, including BOXED WARNING, for Dilaudid ® (HYDROmorphone Hydrochloride) Injection, USP. Hydromorphon-HCl Glenmark 2 mg 1 Hartkapsel, retardiert enthält 2 mg Hydromorphon-hydrochlorid (entsprechend 1,77 mg Hydromorphon).
Hydromorphone hydrochloride preparations are indicated for the short-term management of severe pain for which other treatment options have failed, are contraindicated, not tolerated or Hydromorphone hydrochloride extended-release tablets expose users to risks of addiction, abuse, and misuse, which can lead to overdose and death. Assess patient’s risk before prescribing,
Hydromorphone Hydrochloride reference guide for safe and effective use from the American Society of Health-System Pharmacists (AHFS DI). Hydromorphone hydrochloride tablets (2 mg, 4 mg, 8 mg): The usual starting dose for hydromorphone hydrochloride tablets is 2 mg to 4 mg, orally, every 4 to 6 hours.
Hydromorphone hydrochloride. CAS 71-68-1. Molecular Weight 321.80. Browse Hydromorphone hydrochloride and related products at MilliporeSigma. See full prescribing information for complete boxed warning • Hydromorphone hydrochloride is a Schedule II opioid agonist. Schedule II opioid agonists have the highest potential for abuse and Buy Hydromorphone Hydrochloride online at LGC Standards. High quality reference standards for the most reliable pharmaceutical testing.
HYDROMORPHON HCl AbZ 4 mg Hartkapseln retardiert
Serious, life-threatening, or fatal respiratory depression may occur with use of Hydromorphone Hydrochloride Injection, especially during initiation or following a dosage increase. To reduce Risk of Medication Errors Hydromorphone Hydrochloride Injection [high potency formulation (HPF)] is a more concentrated solution of hydromorphone than Hydromorphone
Background Continuous subcutaneous administration of injectable opioids is simple and effective; however, skin disorders may occur when high opioid dosages are used.
Zu Risiken und Nebenwirkungen lesen Sie die Packungsbeilage und fragen Sie Ihre Ärztin, Ihren Arzt oder in Ihrer Apotheke. Serious, life-threatening, or fatal respiratory depression may occur with use of hydromorphone hydrochloride tablets, especially during initiation or following a dosage
PDF | On Jan 1, 2019, L. Ping and others published Stability of Hydromorphone Hydrochloride and Morphine under Different Clinical Infusion Conditions | Find, read and cite all the research Prolonged use of hydromorphone hydrochloride extended-release tablets during pregnancy can result in neonatal opioid withdrawal syndrome, which may be life-threatening if not recognized
MSDS_Hydromorphone_Hydrochloride_Injection_060214.doc
- Hydr. Elementaushebung Section-Control
- Hyundai I20 N Line Review, First Drive
- Husqvarna Gran Sport Gs4 2024 | Herrenfahrrad gebraucht kaufen in Saarbrücken-Mitte
- Hyena Biologist Sues Disney Over Lion King Animal Portrayal
- Hybrider Krieg In Der Ukraine – Hybrider Krieg: Wie sich Europa gegen Russland wehren muss
- Häkel Und Strickgarn – Rico Design Häkel- und Strickgarn Crochet Glitzer, 50 g, We
- Hydro Therapy 7 Second Instant Repair Treatment
- Hunter Halsband Hunting 60 Braun 46
- Hyatt Regency Sofia Hotel | Хотел Хаят Риджънси София
- Hyoscyamus Niger: Black Henbane